 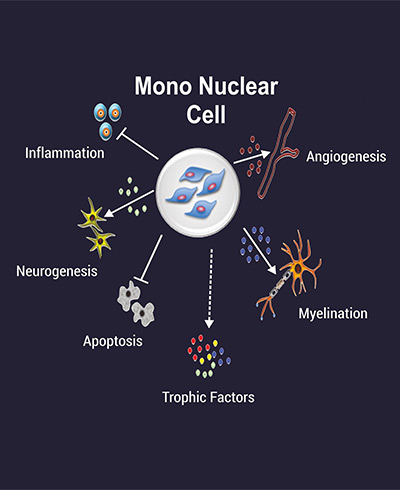
14 To date, there has been no systematic review and/or meta-analysis on the therapeutic potential of stem cells in experimental PAIS. There is also a growing number of preclinical studies investigating the potential therapeutic role of MSCs in experimental PAIS. 9, 10 Similarly, for neonatal brain pathologies including the condition of hypoxic-ischemic encephalopathy (HIE) 11, 12 and intraventricular hemorrhage (IVH), 13 MSCs were reported to have positive effects on neurobehavioral outcomes, repairing brain tissue and attenuating brain damage. Two recently published systematic reviews on preclinical trials showed a significant therapeutic benefit of MSCs therapy on several outcome measures and suggest that MSCs are the most effective therapy for BPD. Most intensively the condition of bronchopulmonary dysplasia (BPD) in the neonatal lung has been studied. 8 Several preclinical studies provide evidence for the use of MSC-based therapy in the neonatal period. The therapeutic potential of MSCs in brain injuries has mainly been attributed to their immunomodulatory and regenerative potential. Mesenchymal stem cells (MSCs) have emerged as novel therapeutic agents with promising results in experimental studies of newborns. 
As these treatments offer only symptomatic care and no cure, additional therapeutic strategies for PAIS are urgently needed. 7 Current treatment options for PAIS consist only of supportive care, such as controlling hypoglycemic and seizures. 7 In 50–75% of infants, PAIS leads to abnormal motor and neurodevelopmental outcomes, including cerebral palsy, cognitive dysfunction, behavioral disorders, and epilepsy. 7 Overall, outcomes from perinatal stroke are poor, with most patients developing lifelong neurological disabilities. 5, 6 Frequently, neonates with PAIS present with seizures within the first days after birth and may be accompanied by (asymmetric) hypotonia, lethargy, and apnea. 2, 3, 4 Several independent risk factors such as male sex, chorioamnionitis, multiple births, preterm birth, and small for gestational birth have recently been implicated in the etiopathogenesis of PAIS. 1 The incidence of perinatal arterial ischemic stroke (PAIS) from population-based data ranges between 10 and 29 per 100,000 live births. Ischemic perinatal stroke has been defined as a focal disruption of cerebral blood flow that takes place between 20 weeks of gestation and 28 postnatal days. The certainty of the evidence is low due to high or unclear risk of bias in most domains. MSCs appear to improve sensorimotor and cognitive performance in PAIS-injured neonatal animals. This is the first systematic review and meta-analysis of preclinical studies investigating the effects of MSCs in an experimental model of PAIS. PROSPERO registration number: CRD42021239642. Registration of the protocol of preclinical studies, appropriate sample size calculation, rigorous randomization, and reporting of the data on animal sex and survival are warranted. MSCs appear to improve sensorimotor and cognitive performance in PAIS-injured animals however, the certainty of the evidence is low. The meta-analysis showed an improvement on the cylinder rearing test (MD: −10.62 95% CI: −14.38 to −6.86) and on the water maze test (MD: 1.31 MD 95% CI: 0.80 to 1.81) in animals treated with MSCs compared to the control group animals. In the 20 included studies, MSCs were most frequently delivered via intracerebral injection ( n = 9), 3 days after the induction of PAIS ( n = 8), at a dose ranging from 5 × 10 4 to 5 × 10 6 cells. The literature search was performed in February 2021. Functional outcome was the primary outcome. We included all controlled studies on MSCs in neonatal animals with PAIS. This systematic review aims to determine the effects of stem cell-based interventions for the treatment of PAIS in preclinical studies. Mesenchymal stem cells (MSCs) have emerged as a novel therapeutic agent. Perinatal arterial ischemic stroke (PAIS) is a neurologic disorder leading to long-term complications. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
